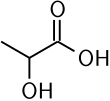
Lactic Acid
This is a naturally occurring acid in milk. It is often used to produce pH levels like those of the hair and skin, but has a more valuable purpose in that it aids the Natural Moisturizing Factor (NMF) of the hair and skin.
Lactic Acid is a powerful alpha hydroxy acid, commonly derived from milk, as well as fruits, vegetables, and other plants. It is commonly believed to be more “gentle” than glycolic acid, while still providing effective exfoliation.
Lactic acid works by helping to slough off the outer “dead or damaged” layer of skin cells, speeding up the rate of cell turn over, and helping to diminish common skin concerns such as fine lines and wrinkles, as well as acne and skin discoloration. It also hydrates the skin, and increases natural barrier lipids in the outer layer of the skin.
When using lactic acid, or any alpha hydrocy acid, skin is more sensitive to the sun, so it’s even more important to protect your skin by using broad-spectrum sun protection – or avoiding sun exposure.
Lactic acid is an organic acid. It has a molecular formula CH3CH(OH)COOH. It is white in the solid state and it is miscible with water. When in the dissolved state, it forms a colorless solution. Production includes both artificial synthesis as well as natural sources. Lactic acid is an alpha-hydroxy acid (AHA) due to the presence of a hydroxyl group adjacent to the carboxyl group. It is used as a synthetic intermediate in many organic synthesis industries and in various biochemical industries. The conjugate base of lactic acid is called lactate (or the lactate anion). The name of the derived acyl group is lactoyl.
|
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Hydroxypropanoic acid | |||
Other names
| |||
| Identifiers | |||
| |||
3D model (JSmol)
|
|||
| 3DMet | |||
| 1720251 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.017 | ||
| EC Number |
| ||
| E number | E270 (preservatives) | ||
| 362717 | |||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII |
| ||
| UN number | 3265 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H6O3 | |||
| Molar mass | 90.078 g·mol−1 | ||
| Melting point | 18 °C (64 °F; 291 K) | ||
| Boiling point | 122 °C (252 °F; 395 K) at 15 mmHg | ||
| Miscible | |||
| Acidity (pKa) | 3.86, 15.1 | ||
| Thermochemistry | |||
Std enthalpy of
combustion (ΔcH⦵298) |
1361.9 kJ/mol, 325.5 kcal/mol, 15.1 kJ/g, 3.61 kcal/g | ||
| Pharmacology | |||
| G01AD01 (WHO) QP53AG02 (WHO) | |||
| Hazards | |||
| GHS labelling: | |||
 | |||
| H315, H318 | |||
| P280, P305+P351+P338 | |||
| Related compounds | |||
Other anions
|
Lactate | ||
Related carboxylic acids
|
|||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
In solution, it can ionize by loss of a proton to produce the lactate ion CH
3CH(OH)CO−
2. Compared to acetic acid, its pKa is 1 unit less, meaning lactic acid is ten times more acidic than acetic acid. This higher acidity is the consequence of the intramolecular hydrogen bonding between the α-hydroxyl and the carboxylate group.
Lactic acid is chiral, consisting of two enantiomers. One is known as L-lactic acid, (S)-lactic acid, or (+)-lactic acid, and the other, its mirror image, is D-lactic acid, (R)-lactic acid, or (−)-lactic acid. A mixture of the two in equal amounts is called DL-lactic acid, or racemic lactic acid. Lactic acid is hygroscopic. DL-Lactic acid is miscible with water and with ethanol above its melting point, which is about 16 to 18 °C (61 to 64 °F). D-Lactic acid and L-lactic acid have a higher melting point. Lactic acid produced by fermentation of milk is often racemic, although certain species of bacteria produce solely D-lactic acid.[citation needed] On the other hand, lactic acid produced by anaerobic respiration in animal muscles has the (L) enantiomer and is sometimes called "sarcolactic" acid, from the Greek sarx, meaning "flesh".
In animals, L-lactate is constantly produced from pyruvate via the enzyme lactate dehydrogenase (LDH) in a process of fermentation during normal metabolism and exercise. It does not increase in concentration until the rate of lactate production exceeds the rate of lactate removal, which is governed by a number of factors, including monocarboxylate transporters, concentration and isoform of LDH, and oxidative capacity of tissues. The concentration of blood lactate is usually 1–2 mM at rest, but can rise to over 20 mM during intense exertion and as high as 25 mM afterward. In addition to other biological roles, L-lactic acid is the primary endogenous agonist of hydroxycarboxylic acid receptor 1 (HCA1), which is a Gi/o-coupled G protein-coupled receptor (GPCR).
In industry, lactic acid fermentation is performed by lactic acid bacteria, which convert simple carbohydrates such as glucose, sucrose, or galactose to lactic acid. These bacteria can also grow in the mouth; the acid they produce is responsible for the tooth decay known as caries. In medicine, lactate is one of the main components of lactated Ringer's solution and Hartmann's solution. These intravenous fluids consist of sodium and potassium cations along with lactate and chloride anions in solution with distilled water, generally in concentrations isotonic with human blood. It is most commonly used for fluid resuscitation after blood loss due to trauma, surgery, or burns.